Pigeons have it. Bees have it. Termites, turtles, lobsters and even bacteria have it. And as it recently turns out, humans do too – magnetic properties. Or, rather, equipment that can perceive magnetism, or is itself magnetic. Known as “magnetoception” or “biomagnetism,” it is a highly specialized adaptation that has evolved primarily for navigation.
As you may already know, the Earth generates its own magnetic field via the action of the electric currents in its liquid outer core. The core itself is actually hot enough to randomize the spins of iron, which ordinarily would diminish the material’s magnetism. However, the Earth’s planetary rotation coupled with the convection of the molten iron reorganizes these spins/electrical currents along a polar north-south axis, much like (but in the Earth’s case, not exactly like) a bar magnet. Moreover, when a conducting fluid (namely, the molten iron) flows across an established magnetic field, more electric currents are induced, another magnetic field forms, and so forth. Thus, the original magnetic field is reinforced by this “dynamo”, a phenomenon better formalized as the “Dynamo Theory,” thereby sustaining the Earth’s magnetic field (1).
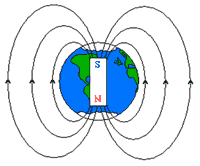
And of course, humans have learned to apply this concept in the creation of compasses, whose magnetic needle will align itself with the natural poles and provide us with direction. Similarly, many animal phyla have developed means to exploit this magnetic field for orientation themselves, especially with migratory behavior. And although an ever-growing number of animals exhibit some degree of magnetic sensitivity (in fact, Google Satellite has revealed that even cows and deer orient themselves along the magnetic axis!) (2), little research has been conducted on the mechanism by which magnetoception occurs. Of the well-studied models, three theories have been established: internal magnetite, cryptochrome detection, and inductive sensing.
Magnetite is a type of iron oxide mineral, and it is the most magnetic naturally occurring mineral. Apparently, not only can many animals biologically synthesize it, but they can also do so in an unusual crystal conformation: prismatic, unlike the octahedra form seen commonly with geological magnetite (3). This shape is a clever biological contrivance to optimize the magnetic moment per particle, a measure of a magnet’s proclivity to align with a magnetic field. Bees have magnetite deposited along the cell membranes of certain neurons, and these neurons may become depolarized by induction when exposed to a certain magnetic field alignment. Homing pigeons and rainbow trout contain magnetite in the back of their beaks and noses, respectively. Both animals come equipped with a system that can signal back the directional information from the magnetite particles to the brain via the trigeminal nerve. Certain bacteria and fungi precipitate magnetite or greigite (another magnetic iron oxide) intercellularly, and with this they affix themselves along Earth’s magnetic field lines (4). Even chiton grow dental magnetite, using them as teeth to cling to and scrap food from rocks.

Even humans possess magnetite traces throughout the brain, including in the cerebellum and meninges (3). Its role in us remains unclear; in fact, it may serve no purpose at all, given its diffuse distribution and lack of an associated organ or system to regulate or interpret a magnetic reading. At least, no such purpose has been discovered yet. Some research has noted higher levels of magnetite in Alzheimer’s (11), but why this is or if there is a direct connection eludes us. Others suggest a weak magnetic sense may exist within us, though (7).
Another method used by animals to perceive and orient to Earth’s magnetic field is through specific light sensitive proteins called cryptochromes. Several bird species, including European robins, fruit flies, and even some plants (whose response is not entirely described below) are examples that use these cryptochromes to detect the magnetic field (12). How cryptochromes work is a bit more complicated, and several models have been proposed in understanding its mechanics. One model suggests that when a cryptochrome is hit by blue light, it experiences a change in which it becomes two radicals. Radicals are molecules with a single unpaired electron which make it more reactive. The unpaired electrons in the radical pair have entangled spins that are influenced by the magnetic field. This interaction between the electron spins and the magnetic field will in turn determine how long the cryptochrome stays in an activated state, thereby affecting the animal’s light receptors and ultimately visual perception. It is believed, then, that the birds and fruit flies can actually “see” the magnetic field so to speak, whereby visual modulation by the magnetic field occurs with different head positions, and in this fashion they can localize according to the Earth’s magnetic field (5,6).

Finally, magnetoception happens by yet another means, albeit less directly: inductive sensing. Cartilaginous fish such as sharks and stingrays best use inductive sensing for both navigation and prey capture, controlled by the ampullae of Lorenzini. Ampullae of Lorenzini are special electromagnetic sensing organs along the head and nose that form a lattice of mucus-filled canals, connecting an outer pore to an internal electroreceptor. Natural electromagnetic fields arise from external, ionospherical, and magntospherical electrical current systems, and by the dynamo relationship of ocean currents with the earth’s magnetic field (8). The organs sense a voltage difference between the pore and electroreceptor base, and this voltage difference affects the rate of the nerve activity. With this input they can not only localize prey (and how this is performed deserves its own article), but they can infer their magnetic heading as well (9,10).

Despite a recent surge of research exploring this remarkable ability, magnetoception continues to be the most poorly understood natural sense. And in light of its mystery, it is exciting to realize how pervasive this capacity is in such a range of animals, from simple bacteria to complex humans. Magnetoception holds promising insight for many ethological topics, including migratory behavior in response to seasonal change, breeding patterns, and foraging. And in humans, who knows… perhaps research will yield a sixth sense unbeknownst to us!
_________________________________________________________________________________
References
1.Phillips, Tony (December 29, 2003). “Earth’s Inconstant Magnetic Field”. Science@Nasa. http://science.nasa.gov/science-news/science-at-nasa/2003/29dec_magneticfield/. Retrieved September 21, 2010.
2. Burda, Hynek; Sabine Begall, Jaroslav Cerveny, Julia Neef, and Pavel Nemec. 2009. “Extremely low-frequency electromagnetic fields disrupt magnetic alignment of ruminants”. Proceedings of the National Academy of Sciences 106 (14): 5708–13.
3. Kirschvink, JL; Kobayashi-Kirschvink, A; and Woodford, BJ. 1992. “Magnetite biomineralization in the human brain.” Proc Natl Acad Sci U S A. 89(16): 7683–7687.
4. Bazylinski, Dennis. 1995. “Controlled biomineralization of magnetic minerals by magnetotactic bacteria”. Chemical Geology 132: 191.
5. Wiltschko, W and Wiltschko, R. 2008. “Magnetic orientation and magnetoreception in birds and other animals.” Journal of Comparative Physiology. A, Neuroethology, Sensory, Neural, and Behavioral Physiology 191 (8): 675–93.
6. Gegear, Robert J.; Amy Casselman, Scott Waddell, Steven M. Reppert. 2008. “Cryptochrome mediates light-dependent magnetosensitivity in Drosophila“. Nature 454 (7207): 1014–1018.
7. Carrubba, S; Frilot, C; Chesson, AL; Marino, AA. 2007. “Evidence of a nonlinear human magnetic sense”. Neuroscience 144 (1): 356–67.
8. Chave, AL. 1990. “Low-Frequency, Motionally Induced Electromagnetic Fields in the Ocean.” Journal of Geophysical Research 95 (C5): 7185-7200.
9. Murray, RW. 1960. “Electrical sensitivity of the ampullae of Lorenzini”. Nature 187: 957.
10. Fields, RD, Fields, KD, Fields, MC. 2007. “Semiconductor gel in shark sense organs?”. Neurosci. Lett. 426,: 166–170.
11. Hautot, D; QA; N Khan, and Dobson, J. 2003. Preliminary evaluation of nanoscale biogenic magnetite in Alzheimer’s disease brain tissue. Proc Biol Sci. 270(Suppl 1): S62–S64.
12. Cryptochrome and Magnetic Sensing, Theoretical and Computational Biophysics Group at the University of Illinois at Urbana-Champaign. Accessed 21 September 2010.


I’ve studied what I believe to be my magnetoreceptive abilities for 3 years. I have found evidence that my psychiatric symptoms are in fact an internal GPS, indicating whether I’m north or south of magnetic home. Check out my website for more information.